Atomic Radius
What is Atomic Radius?
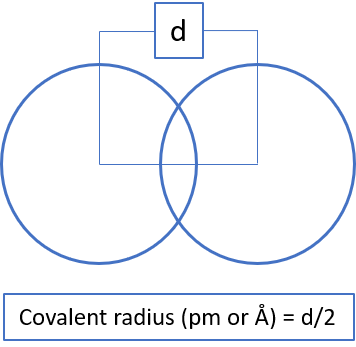
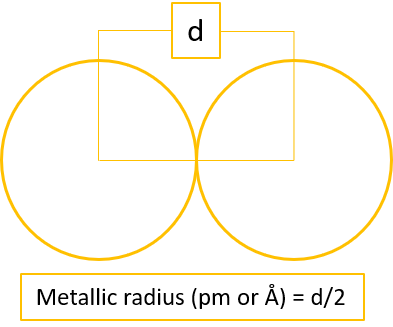
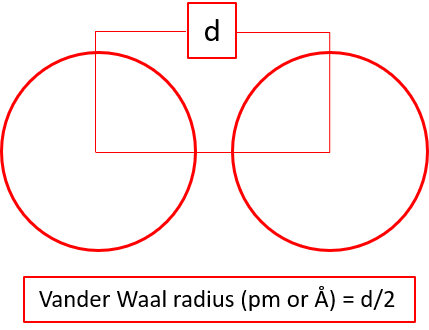
Atomic radius is a measure of atomic size. There are three different ways in which atomic radius can be calculated; covalent radius, metallic radius,
and Vander Waal radius. The atomic radius is calculated using X-ray diffraction method.
Types of Atomic Radius
Applications of Atomic Radius
The atomic radius finds application in the field of material science:
- One of the important applications of the atomic radius is in interstitial compounds such as transition metal borides, nitrides, and carbides.
- Transition metal atoms can be spherically packed with smaller atoms, such as carbon, nitrogen, boron, etc., in their octahedral or tetrahedral
interstices or voids. - The ratio of the atomic radius of the smaller atom to that of the transition metal is important for predicting the stability of a transition metal
boride, carbide, etc. - Transition metal borides are ultra-high-temperature ceramics that can be used in hypersonic vehicles such as spacecraft. When a spacecraft from
space reenters the earth’s atmosphere, it gets heated up to a very high temperature of 1700°C; therefore, ultra-high temperature ceramic is needed
to withstand that temperature. - A hypersonic vehicle can travel four times faster than the speed of sound and helps launch a satellite at a low cost.
- The transition metal carbide is used as a hard coating to protect the surface of cutting tools from wear and erosion, resulting in their extended life.
Groupwise Analysis
Note: It is to be noted covalent radius is considered for nonmetals, metallic radius is considered for metals, and VanderWaal radius is considered for noble gases for the trend analysis. However, sometimes when the data of respective radius is unavailable, then other type of radius is taken into consideration for such elements.
References:
- Beatriz Cordero, Verónica Gómez, Ana E Platero-Prats, Marc Revés, Jorge Echeverría, Eduard Cremades, Flavia Barragán, and Santiago Alvarez. Covalent radii revisited. Dalton Transactions, pages 2832, 2008. URL, doi:10.1039/b801115j.
- William M Haynes. CRC Handbook of Chemistry and Physics. 100 Key Points. CRC Press, London, 95th edition, 2014. ISBN 9781482208689. URL.
- Kyle & laby tables of physical & chemical constants. (2017). 3.7.5 atomic radii. [Online; accessed 30-April-2017]. URL.
- https://www.rsc.org/periodic-table/
Periodwise Analysis
Note: It is to be noted covalent radius is considered for nonmetals, metallic radius is considered for metals, and VanderWaal radius is considered for noble gases for the trend analysis. However, sometimes when the data of respective radius is unavailable, then other type of radius is taken into consideration for such elements.
References:
- Beatriz Cordero, Verónica Gómez, Ana E Platero-Prats, Marc Revés, Jorge Echeverría, Eduard Cremades, Flavia Barragán, and Santiago Alvarez. Covalent radii revisited. Dalton Transactions, pages 2832, 2008. URL, doi:10.1039/b801115j.
- William M Haynes. CRC Handbook of Chemistry and Physics. 100 Key Points. CRC Press, London, 95th edition, 2014. ISBN 9781482208689. URL.
- Kyle & laby tables of physical & chemical constants. (2017). 3.7.5 atomic radii. [Online; accessed 30-April-2017]. URL.
- https://www.rsc.org/periodic-table/
Transition Metals Analysis
Note: It is to be noted covalent radius is considered for nonmetals, metallic radius is considered for metals, and VanderWaal radius is considered for noble gases for the trend analysis. However, sometimes when the data of respective radius is unavailable, then other type of radius is taken into consideration for such elements.
References:
- Beatriz Cordero, Verónica Gómez, Ana E Platero-Prats, Marc Revés, Jorge Echeverría, Eduard Cremades, Flavia Barragán, and Santiago Alvarez. Covalent radii revisited. Dalton Transactions, pages 2832, 2008. URL, doi:10.1039/b801115j.
- William M Haynes. CRC Handbook of Chemistry and Physics. 100 Key Points. CRC Press, London, 95th edition, 2014. ISBN 9781482208689. URL.
- Kyle & laby tables of physical & chemical constants. (2017). 3.7.5 atomic radii. [Online; accessed 30-April-2017]. URL.
- https://www.rsc.org/periodic-table/
Lanthanide and Actinide Analysis
Note: It is to be noted covalent radius is considered for nonmetals, metallic radius is considered for metals, and VanderWaal radius is considered for noble gases for the trend analysis. However, sometimes when the data of respective radius is unavailable, then other type of radius is taken into consideration for such elements.
References:
- Beatriz Cordero, Verónica Gómez, Ana E Platero-Prats, Marc Revés, Jorge Echeverría, Eduard Cremades, Flavia Barragán, and Santiago Alvarez. Covalent radii revisited. Dalton Transactions, pages 2832, 2008. URL, doi:10.1039/b801115j.
- William M Haynes. CRC Handbook of Chemistry and Physics. 100 Key Points. CRC Press, London, 95th edition, 2014. ISBN 9781482208689. URL.
- Kyle & laby tables of physical & chemical constants. (2017). 3.7.5 atomic radii. [Online; accessed 30-April-2017]. URL.
- https://www.rsc.org/periodic-table/